"In the face of an unprecedented challenge, the JA2 team united government agencies, contractors, academia and industry experts to rewrite the script of our battle against COVID-19." – Mr. Darryl J. Colvin, Joint Program Executive Officer for Chemical, Biological, Radiological and Nuclear Defense (CBRN).
The U.S. held its collective breath this past May as President Biden declared the end of the Nation’s emergency response posture to the relentless COVID-19 pandemic. This announcement marked the culmination of a more than three-year long journey rife with challenges and uncertainties. At the heart of this victory story is the Joint Program Executive Office for Chemical, Biological, Radiological and Nuclear Defense’s (JPEO-CBRND) Joint Assisted Acquisition (JA2) team, whose unwavering dedication paved the way for the delivery of the medical countermeasures to move us out of a pandemic and into the new endemic state with the Coronavirus. This team was called upon to jump in the ring and helped procure more than 2 billion vaccines, 23 million therapeutics doses more than two billion needles and syringes, support for procurement of the President’s 1 billion test kit efforts and support to stand up a domestic medical countermeasure supply chain to protect the Nation from future public health emergencies.
In May 2021, the Defense Department (DOD) signed a memorandum of understanding with the U.S. Department of Health and Human Services (HHS) to support the vaccine development and acquisition of the critical medical countermeasures our country needed to protect our civilians and service members from COVID-19 as the virus swept the globe. JA2 organized and led the acquisition efforts for DOD’s assisted acquisition teams to procure and deliver the medical countermeasures we needed around the globe to confront COVID-19 head-on. The DOD’s primary mission was clear – to provide essential contracting and project management support to tackle the complex challenges posed by the pandemic, orchestrating a cohesive and comprehensive government response. The expertise of the multi-faceted JA2 team comprised various lines of effort, each converging toward the ultimate success of the mission.
 Figure 1 – Diagram of JA2 accomplishments addressing COVID-19 and the future of its efforts in bio-preparedness and defense.
Capacity Expansion: The Backbone of Resilience
Figure 1 – Diagram of JA2 accomplishments addressing COVID-19 and the future of its efforts in bio-preparedness and defense.
Capacity Expansion: The Backbone of Resilience
In a testament to their resilience, the JA2 team spearheaded the Capacity Expansion (CapEx) initiative, ensuring the domestic manufacturing capabilities of critical supplies could keep pace with this pandemic's demands and stood up critical manufacturing capabilities for our domestic medical supply chain at the same time. "Our CapEx team ensured the Nation’s ability to manufacture COVID 19 vaccines to meet global demands. Our program expanded the industrial base for vaccine production by constructing additional filling lines, consumables production capabilities, vials manufacturing, and raw material development to reduce the US’ reliance on OCONUS manufacturing." said, Stefanie Hagan, Joint Product Lead for CapEx. This achievement not only showcased the team's commitment but also contributed to the revival of the country's supply chain, generating an economic ripple effect of over $2 billion that was injected across 20 communities in the U.S. “This initiative not only constructed facilities to manufacture vaccines, vaccine components, fill vials, and packaging, it brought jobs to these communities. These facilities and communities will play a vital role in the event of future pandemics." says Hagan.
The JA2 CapEx team's influence extended to industrial base expansion (IBX) efforts, embarking on medical countermeasure raw materials manufacturing efforts across 22 states and territories. The team help ensure the successful investment of more than $2.4 billion over more than 20 initiatives; infusing the Nation's response capability with enhanced resilience and adaptability to face future crises.
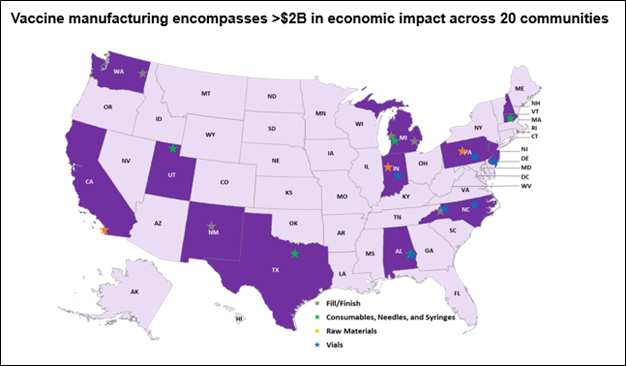 Figure 2 - Map outlining the JA2 Capacity Expansion team's efforts establishing domestic medical countermeasure manufacturing facilities.
Vaccines, Therapeutics, and Enablers: An Unwavering Commitment
Figure 2 - Map outlining the JA2 Capacity Expansion team's efforts establishing domestic medical countermeasure manufacturing facilities.
Vaccines, Therapeutics, and Enablers: An Unwavering Commitment
Arguably the most significant feather in the JA2 team's cap was its instrumental role they played in procuring vaccines, therapeutics and enablers (VTE). The team acted as a conduit for the procurement of a range of vaccines and therapeutic products, facilitating the Nation's access to variant and pediatric boosters, as well as oral antivirals. The tangible impact extended beyond national borders, as the team coordinated the global donation of vaccines and therapeutics.
"With over $35 billion awarded, we successfully procured more than 2 billion vaccine doses and expertly pivoted to award booster dose contracts," proclaimed Drew Joyce, Joint Product Lead for VTE. This numerical testament highlighted the team's responsiveness and dedication to achieving the Nation's vaccination objectives. But what are vaccines if they cannot be administered or monitored for safety and efficacy? The team took this effort over the finish line by ensuring the seamless distribution of vaccines, the JA2 team procured over 1 billion needles and syringes from seven suppliers and delivered vital vaccine monitoring services.
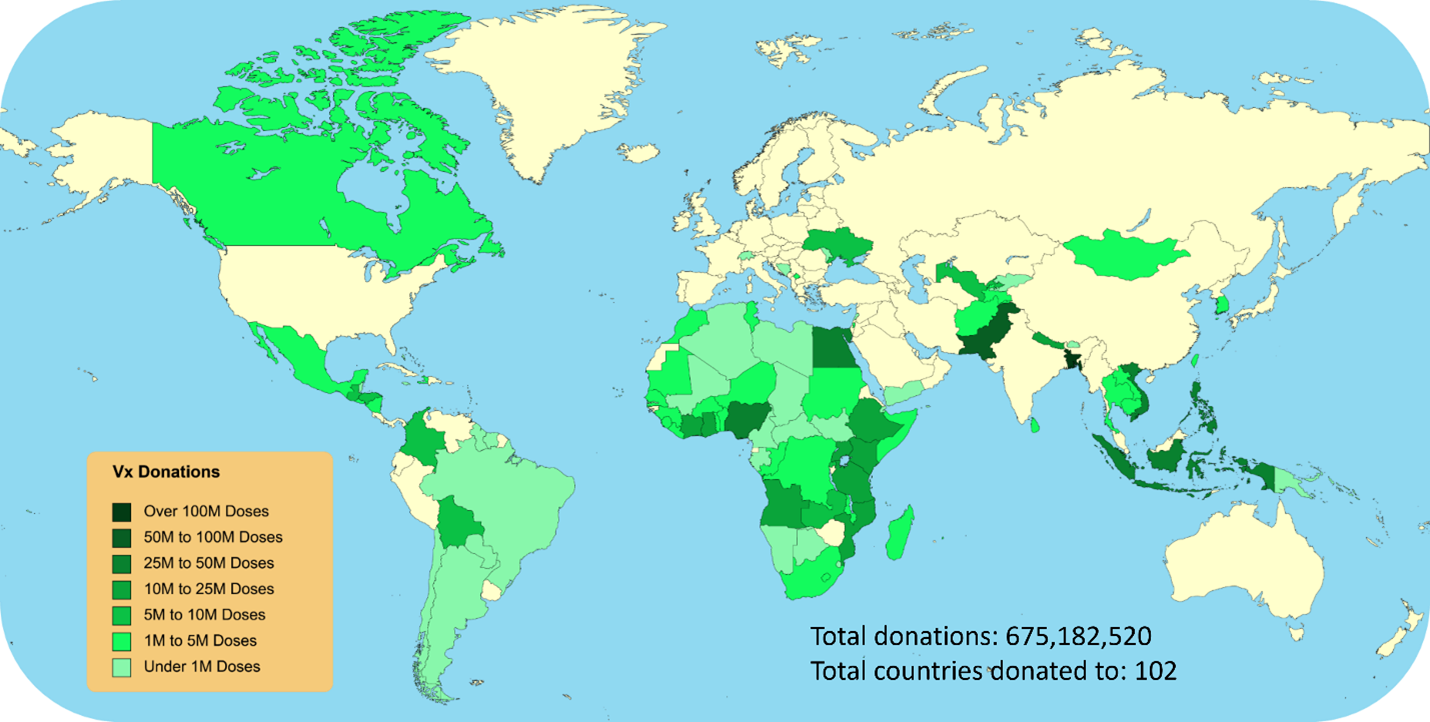 Figure 3 - Map representing vaccine doses donated globally by the JA2 Vaccine, Therapeutics and Enablers team.
Figure 3 - Map representing vaccine doses donated globally by the JA2 Vaccine, Therapeutics and Enablers team.
Screening and Diagnostics: Laying the Foundations
At the core of the JA2 team's achievements was their procurement of screening and diagnostics (S&D) capabilities and the expansion of domestic manufacturing for screening and diagnostic materials. Additionally, the establishment of “Operation ET,” the $650 million school and community COVID testing program for students (kindergarten to 12th grade) and underserved populations served as a fundamental cornerstone to detection of the virus. One of the initiatives was a colossal 10-month effort to procure 910 million over-the-counter COVID-19 rapid antigen tests. These tests, acquired for U.S. Postal Service delivery and storage in the Strategic National Stockpile, exemplified the JA2 team's unswerving dedication to the health and safety of the Nation. These efforts provided the Nation with the necessary tools to identify and combat the virus over the many variant and subvariant waves of the pandemic. "Our strategic direction set the stage for state-level implementation, ensuring comprehensive testing across various geographical regions with 42 U.S. manufacturing facilities built in 22 states and U.S. territories," explained Dr. Mike Ingram, Joint Product Lead for S&D, emphasizing the collaborative nature of the effort.
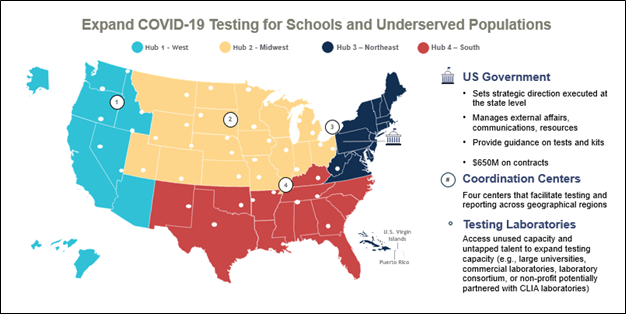 Figure 4 – Map representing the Screening & Diagnostics team's efforts to expand COVID-19 testing for schools and underserved populations on behalf of the U.S. government, depiction shows the regional coordination centers and testing laboratories across the United States.
Figure 4 – Map representing the Screening & Diagnostics team's efforts to expand COVID-19 testing for schools and underserved populations on behalf of the U.S. government, depiction shows the regional coordination centers and testing laboratories across the United States.
The S&D team also successfully helped fulfill the President’s 1 billion test kit commitment. The team expanded production increases every month, allowing the U.S. to create 217 million tests, 1 billion swabs and 1.1 billion pipette tips per month – all essential for creating over-the-counter test kits individuals received in the mail or purchased at pharmacies.
Future Preparedness
The JA2 team’s efforts have left their mark on the future of bio-preparedness. Their efforts not only mitigated the immediate challenges of COVID-19 but created tools and capabilities to address emerging threats. By ensuring a more stable supply chain, cultivating a ready industrial base and fostering seamless coordination among government agencies, contractors, academia and industry experts, the JA2 team’s legacy will empower the nation to confront future crises with unshakable resolve.
“In rewriting our narrative against COVID-19, the JA2 team has laid the groundwork for a more resilient future,” affirmed Nicole Kilgore, Deputy Joint Program Executive Officer for CBRN and lead for the JA2 program. “The tale of JA2’s contributions serve as a beacon of hope and a testament to the transformative potential of collaboration, innovation and forward-thinking in the face of adversity,” says Kilgore.
As of September 30, 2023, the JPEO officially closed out of the MOU and completed the warm handover of the contracting and program management efforts for COVID-19 back to HHS. This is an important milestone for JA2 and the JPEO-CBRND. Over 30 contracts and agreements that helped JA2 achieve victory in the battle against COVID-19 seamlessly transferred from DOD to HHS, highlighting a successful collaboration and strategic transition. The JPEO-CBRND is returning to its traditional portfolio of developing CBRN defense capabilities, including medical countermeasures, for the joint force to keep warfighters safer in CBRN contested environments.
Celebrating Those Who Served
In August 2023, two years after the MOU was originally signed by DOD and HHS, the team gathered to mark the accomplishments and service to their mission. Some traveled to gather in person and many more tuned in remotely to commemorate the occasion. All staff who supported the efforts from across the JPEO-CBRND were recognized for their tireless efforts and contributions to accomplish the mission. Ms. Kilgore presided over the awards ceremony and presented staff with a JA2 certificate of appreciation and a JA2 challenge coin. The most meaningful aspect seemed to be getting the team together to reflect on the somber reason JA2 was stood up as well as the sacrifices and accomplishments from the team over the past three years of the JPEO’s service to the DOD, our Nation and the globe.
 Figure 5 – Deputy JPEO and JA2 Executive Lead, Ms. Nicole Kilgore, and JA2 headquarter staff gathered at the August 2023 awards ceremony.
Figure 5 – Deputy JPEO and JA2 Executive Lead, Ms. Nicole Kilgore, and JA2 headquarter staff gathered at the August 2023 awards ceremony.
Should the Nation need our support again, we stand ready to assist. As a result of the pandemic, we’re a more able team in the face of future biothreats. We’re armed with the lessons, knowledge, and dedication of a workforce whose been tested and proved their ability to operate through adversity and deliver the medical countermeasures our Nation needs.